| Chapter 8: Nucleophilic Substitution |
Reaction of Alcohols with other Halogenating agents (SOCl2,
PX3)
(review of chapter 4)
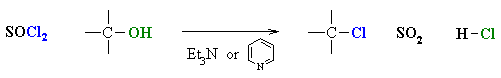 |
 |
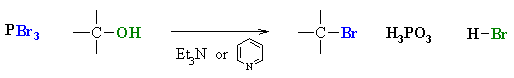 |
Reaction type: Nucleophilic Substitution (SN1 or SN2)
Summary
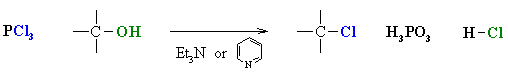
- Alcohols can also be converted to alkyl chlorides using thionyl chloride, SOCl2, or phosphorous trichloride, PCl3.
- Alkyl bromides can be prepared in a similar reaction using PBr3.
- Used mostly for 1o and 2o ROH (via SN2 mechanism)
- In each case a base is used to "mop-up" the acidic by-product.
- Common bases are triethylamine, Et3N, or pyridine, C6H5N.
- In each case the -OH reacts first as a nucleophile, attacking the electrophilic center of the halogenating agent.
- A displaced halide ion then completes the substitution displacing the leaving group.
- Note that it is not -OH that leaves, but a much better leaving group (e.g. SO2)
- The advantage of these reagents is in that the reaction is not under the strongly acidic conditions like using HCl or HBr.
| SN2 MECHANISM FOR REACTION
OF ALCOHOLS WITH SOCl2 |
|
| Step 1: Step 2: Step 3: Step 4: |
 |
Related Reactions
| © Dr. Ian Hunt, Department of Chemistry |