| Chapter 14: Organometallic Compounds |
| Chapter 14: Organometallic Compounds |
Oxymercuration-Demercuration of Alkenes

Summary
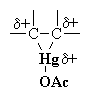

|
|
|
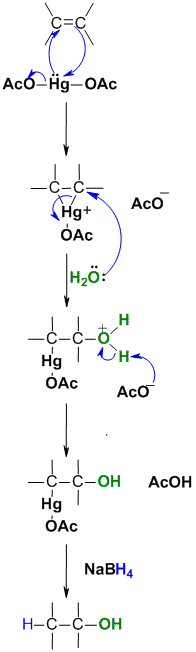
| Step 1:
The C=C π electrons act as the nucleophile with the electrophilic Hg and loss of an acetate ion as a leaving group, forming the cyclic mercurinium ion. |
|
| Step 2:
Water functions as a nucleophile and attacks one of the carbons substituted with mercury resulting in cleavage of the C-Hg bond. |
|
| Step 3:
The acetate ion functions as a base deprotonating the oxonium ion to give the alcohol. This completes the oxymercuration part of the reaction. |
|
| Step 4:(mechanism
not shown) The hydride reduces the Hg off, creating a C-H bond while breaking the C-Hg bond. This is the demercuration part of the process. This step is not stereoselective. |
|
| © Dr. Ian Hunt, Department of Chemistry |