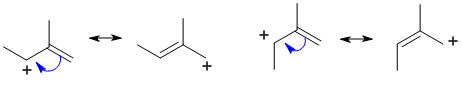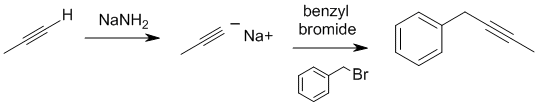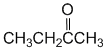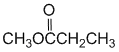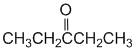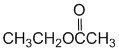Chem 351 Final Fall 2017
Here is an post-mortem analysis / "how to" for
the FINAL. The questions are split by the sections. At the start of each section
are a few suggestions of what to look for or how to tackle the question type.
RELATIVE PROPERTIES:
Identify the controlling feature,
which is not always as obvious as
it may appear. Look for two pairs of similar systems to compare that
have
minimal differences in structure.
| Qu 1: |
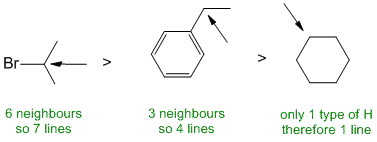
The number of lines = multiplicity (i.e. coupling) of the signals for each of the positions indicated is determined by the number of neighbours (n) that are of a different type (since H of the same type do not show coupling e.g. the CH2 groups in cyclohexane (where there is only 1 type of H). |
 |
| Qu 2: |
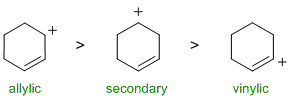
A question about stability carbocations. Remember that the order of stability is tertiary > secondary > primary and that allylic carbocations are further stabilised by resonance while vinyl cations are less stable than primary cations. The secondary allylic carbocation is the most stable (due to resonance delocalisation) and the vinyl carbocation is the least stable.
|
 |
| Qu 3: |
Really a question about alkene stability. Notice they are isomeric (C6H12). The more stable the alkene the less exothermic (more negative) it's heat of combustion. In terms of alkene stability, the general rule is that the more highly substituted the alkene (i.e. more attached alkyl groups) then the more stable the alkene. |
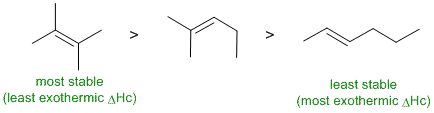 |
Qu 4: |
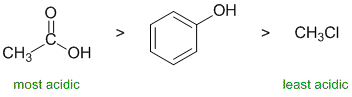
Acidity.... Need to look at the stability of the conjugate bases and consider the factors that affect that stability. Remember that the lower the pKa the stronger the acid. Here we have a phenol (pKa about 10), chloromethane and a simple carboxylic acid (pKa about 5). A carboxylic acid is more acidic than a phenol because the conjugate base (the carboxylate) delocalises the -ve charge to a second electronegative oxygen atom. In chloromethane, the H is attached to a C and therefore is not going to be particularly acidic... A knowledge of the pKa values of common acids really helps! |
 |
Qu 5: |
Basicity.... the strongest base will produce the most the greatest amount of the conjugate base. The strongest base is the ammonia (associated pKa = 10) then water (pKa = 0) and lastly the methane (pKa "very -ve").... for basicity, think about where the e are coming from ? N and O both have lone pairs while the C in methane does not. Students often make an error because they evaluate the basicity of -CH3 rather than CH4. |
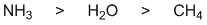 |
Qu 6: |
IR spectroscopy : stretching vibrations are affected by bond strength and the masses of the attached atoms (Hooke's law model). Lighter atoms (e.g. H) give higher frequencies (e.g. C-H) and stronger bonds give higher frequencies (e.g. C=C > C-O). |
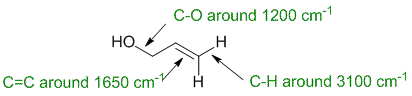 |
Qu 7: |
Chemical shifts of the groups in question in these systems are determined by the position of the H (that is what is changing). Chemical shifts are affected by electronegative atoms (especially such as O) and magnetic anisotropy (due to pi systems). The carboxylic acid H (12ppm) is deshielded by the electronegative O and the acidic character of the group). The methyl -CH3 group is affected by the benzene ring (about 2.2 ppm) and the methylene -CH2- is affected by both the benzene ring and also by the proximity of the acid group (about 3.5 ppm). |
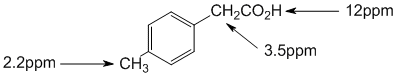 |
| Qu 8: |
All about the nucleophilicity of these molecules. All are negative, and we have two O systems and one S. If we first compare the two O systems, we have a carboxylate and an alkoxide. The resonance delocalisation in the carboxylate makes the lone pairs on the O less available and hence less nucleophilic. Now for the S vs the alkoxide O (which it is most like). S is below O in the periodic table and the increased size and hence polarisability of large atoms makes them more nucleophilic. |
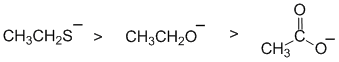 |
| Qu 9: |
This is a radical substitution reaction of alkanes (i.e. sp3 C-H bonds), remember the laboratory experiment ? For chlorination, the reaction is controlled by a combination of the relative reactivity of the radical formed and the number of H that yield that radical. The relative reactivity of tertiary : secondary : primary systems are 5.2 : 3.9 : 1. The calculations are outlined on the figure on the starting material. |
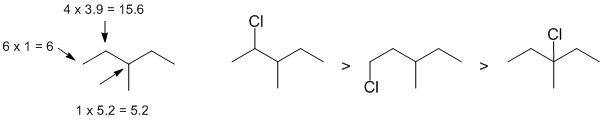 |
| Qu 10: |
At pH 8, the major species would have the phenol group protonated and the more acidic carboxylic acid deprotonated. There would also be a smaller amount where both are deprotonated. The acidity of the groups (carboxylic acid > phenol) means the final form is very, very unlikely. |
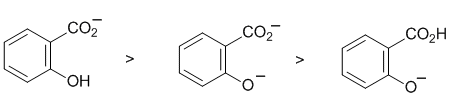 |
| Qu 11: |
Mass spectrometry... 2-chloropropane will give the molecular ion, m+, at 78 g/mol. The peak at 79 (due to the presence of a single atom of 13C, 1.1% natural abundance) will be quite small while the peak at 80 (m+2, due to the 37Cl isotope) will be about 1/3 the height of the molecular ion. |
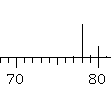 (the image shows the actual MS) (the image shows the actual MS) |
| Qu 12: |
Counting types of hydrogen |
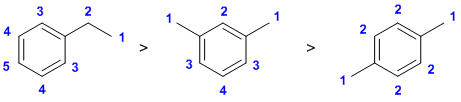 |
MOLECULAR PROPERTIES
No real method here, really just do you
know various aspects of molecular structure / reactions, applied to each of the questions.
| Qu13: |
The major product will be the thioether II because thiols are quite acidic (pKa about 10) and ethoxide is basic (associated pKa = 15) so RSH + RO- gives RS- + ROH and the S system is a far better nucleophile. |
| Qu14: |
The chemical shift of the aldehyde H (9-10 ppm) is due to the magnetic anisotropy of the double bond and the position of the H atom in the deshielding cone of the induced magnetic field. |
| Qu15: |
The reaction is an acid / base reaction and phenols II are more acidic than simple alcohols. Phenols are more acidic due to the resonance delocalisation of the -ve charge from the O on to the C atoms of the benzene ring. |
| Qu16: |
Method I is preferred since the primary nature of both components will facilitate the desired SN2 reaction. Method II will only give an acid / base reaction. |
| Qu17: |
Method I is preferred since method II will result in an E1 where rearrangement will give a different product. |
| Qu18: |
Push the curly arrows! Two of the choices are resonance contributors (check overall charge and changes to sigma bonds). |
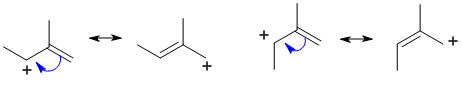 |
REACTIONS:
Three types of questions.
For those
with starting materials work from
the starting materials towards the products using the reagents to "see"
what product to look for.
For those with the product work
backwards....
looking at the functional groups in the products to think about how you
may have got there.
For those wanting reagents look at
the functional groups in the
starting material and products
to try to determine what may have happened. Look at the reagents in
each
option to see what effect they would have on the SM....
| Qu19: |
We should work forwards...Sodium amide will react as a base with the terminal alkyne to create the nucleophilic acetylide ion with will the react via a nucleophilic substitution with the benzyl bromide to give an internal alkyne. Remember to count C atoms. |
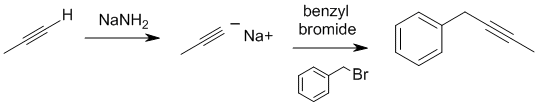 |
| Qu20: |
We should work backwards... The required alkene is the less highly substituted (anti-Zaitsev) product and therefore simple acid dehydration (E1) is not going to work. Conversion of the alcohol to the alkyl halide via a nucleophilic substitution and then elimination using a strong bulky base will give the desired product via an E2
elimination.
|
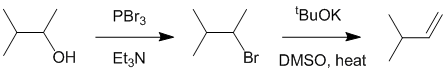 |
| Qu21: |
We should work backwards... An ethyl ether has been made via a Williamson ether synthesis using ethoxide so we need to identify the alkyl halide. The alkyl chloride has been made via a radical substitution of a benzylic position. |
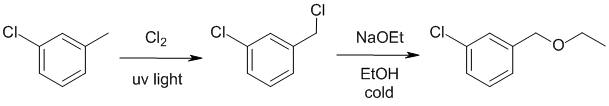 |
| Qu22: |
We should work forwards ...an alcohol to the more highly substituted Zaitsev alkene can be achieved by formation of a chloride thionyl chloride via a nucleophilic substitution and then elimination via an E2
elimination followed by allylic radical substitution would give the desired allylic bromide. |
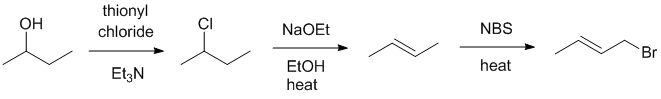 |
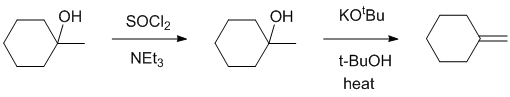
| Qu23: |
We should work backwards... The required alkene is the less highly substituted (anti-Zaitsev) product and therefore simple acid dehydration (E1) is not going to work, but conversion of the alcohol to the alkyl halide using thionyl chloride via a nucleophilic substitution and then elimination using a strong bulky base will give the desired product via an E2
elimination. |
|
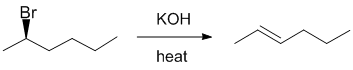
| Qu24: |
We should work forwards...KOH will react with an alky bromide when heated to undergo a Zaitsev E2
elimination to give the more stable, more highly substituted trans-alkene. |
 |
| Qu25: |
We can work forwards... alkane to alkyl bromide via a radical substitution. |
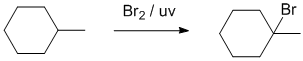 |
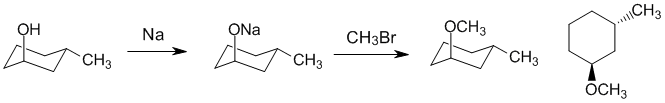
| Qu26: |
We should work backwards.... A methyl ether has been made via a Williamson ether synthesis using methyl bromide so we need to identify the alcohol. Since we are not changing the C-O bond to the ring, the alcohol needs to have the same configuration as the product ether. |
 |
CONFORMATIONAL ANALYSIS:
Understanding the terminology and the
energies involved in
conformational
analysis.
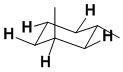
Qu 27:
Draw out the most stable conformation of trans-1,3-dimethylcyclohexane... this will have the two methyl groups on opposite faces of the ring with 1 equatorial which means there are 6 - 1 = 5 equatorial H.
Qu 28:
2-chloro-3-methylbutane is a C5 system, (CH3)2CHCH(Cl)CH3. We need to find the structures that are (a) staggered and with (b) a methyl group anti to the Cl and gauche to a methyl group and (c) a methyl group gauche to Cl and anti to a methyl group

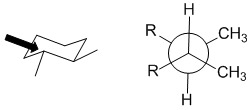
Qu 29:
The torsional angle between the two methyl groups is 60 degrees in this chair conformation of trans-1,2-dimethylcyclohexane.
Qu30:
The torsional angle between the chlorine atoms is 120 degrees in this eclipsed conformation.
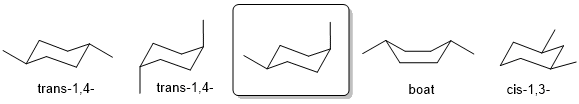
Qu31:
Cis requires that the two methyl groups are on the same face of the ring. The most stable conformation of cis-1,4-dimethylcyclohexane will be a chair conformation with one equatorial and one axial methyl group at opposite ends of the ring.
Qu32:
Since we are looking at a cyclohexane where the two C-C bonds are within the ring structure itself, the best term is gauche. Gauche requires that the two bonds are staggered and at 60 degrees..
Qu33:
The structures are isomeric (same MF) but what type of isomer are they? Comparing the drawings or using models and comparing allows one to determine that the aldehyde and alcohol substituted C both have the opposite configurations and hence they are best described as enantiomers.
Qu34:
Cyclohexane bonds angles are very close to the ideal tetrahedral value of 109.5 degrees as so it has minimal angle strain. Smaller rings (including those in polycyclic systems) will have bond angles with significant deviations from that optimal angle and therefore higher angle strain. Cyclopentane and cycloheptane rings are almost angle strain free, but cyclohexane rings best and are typically said to be "strain free".
SPECTROSCOPY:
Use any IR information to get the
functional groups. Use the H-NMR
to get the number of types of H, how many of each type from the
integral
and what they are next to from the coupling patterns. Chemical shifts
should
tell you if the group is near -O- or maybe C=O groups etc.
| Qu 35: |
IR shows a carbonyl (i.e. C=O) at 1718 cm-1 which is typical for a ketone as is the C-NMR peak at 209 ppm. The H-NMR has 3 types of H. The 3H triplet at 1.1 ppm,
suggests a -CH3 group next to a CH2, the 3H singlet at 2.1 ppm an isolated -CH3 group and the 2H quartet at 2.4ppm a CH2 next to a -CH3 . These pieces of data are consistent with a ketone with a methyl group and an ethyl group : butan-2-one. |
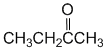 |
| Qu36: |
IR shows a carbonyl (i.e. C=O) at 1741 cm-1 and in the C-NMR (at 175 ppm) which suggests a carboxylic acid or derivative. The H-NMR has 3 types of H. The 3H triplet at 1.2 ppm,
suggests a -CH3 group next to a CH2, the 2H quartet at 2.3 ppm suggests a CH2 next to a -CH3 and the 3H singlet at 3.7 ppm an isolated and deshielded -CH3 group, possibly -OCH3. These pieces of data suggest a methyl ester of propanoic acid. |
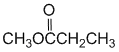 |
| Qu37: |
IR shows a carbonyl (i.e. C=O) at 1716 cm-1 which is typical for a ketone as is the C-NMR peak at 212 ppm. The H-NMR has 2 types of H. The 3H triplet at 1.1 ppm,
suggests a -CH3 group next to a CH2, and the 2H quartet at 2.4ppm a CH2 next to a -CH3 . These pieces of data are only consistent with a symmetrical ketone, pentan-3-one. |
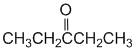 |
| Qu38: |
IR shows no carbonyl (i.e. C=O) near 1715 cm-1 or in the C-NMR (near 200ppm). The C-NMR has 5 types of C, 3 of which (114, 121 and 149 ppm) are in the aromatic region. The H-NMR has 3 types of H. The 3H triplet at 1.4 ppm,
suggests a -CH3 group, the 2H quartet at 4.0 ppm a deshielded CH2 next to a -CH3 and the 2H multiplet at 6.9 ArH. Given the structures, looks like we need a symmetrical system (double the integrations) to have a disubstituted aromatic. If there only 3 ArC types, then it will have to be ortho. |
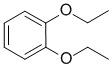 |
| Qu39: |
IR shows a carbonyl (i.e. C=O) at 1743 cm-1 and in the C-NMR (at 171 ppm) which suggests a carboxylic acid or derivative. The C-NMR has 4 types of C. The H-NMR has 3 types of H. The 3H triplet at 1.3 ppm,
suggests a -CH3 group next to a CH2, the 3H singlet at 2.1 ppm an isolated -CH3 group and the 2H quartet at 4.1 ppm a deshielded CH2 next to a -CH3, likely an -OCH2CH3. |
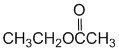 |
| Qu40: |
IR shows a carbonyl (i.e. C=O) at 1712 cm-1 and -OH at 2700-3300 cm-1. The C=O in the C-NMR is at 181 ppm which suggests a carboxylic acid or derivative. The H-NMR has 4 types of H. The 3H triplet at 1.0 ppm,
suggests a -CH3 group next to a CH2, the 2H sextet at 1.7 ppm suggests an -CH2 group with 5 neighbours, the 2H triplet at 2.3 ppm a CH2 next to a -CH3 and the 11.5ppm exchangeable H indicates a carboxylic acid. These pieces of data are consistent with butanoic acid. |
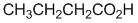 |


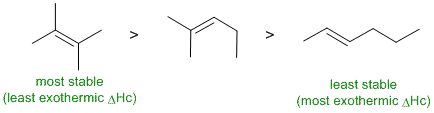



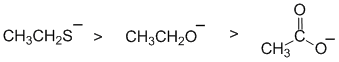


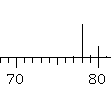 (the image shows the actual MS)
(the image shows the actual MS)