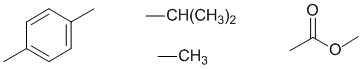Part 8: SPECTROSCOPY
The following data is available from the
question.
Note
: Remember to cross
reference things to confirm.... e.g.
IR may show C=C, use NMR to
confirm that etc.
MS: M+
seen at 178 g/mol. No isotope patterns for Cl or Br.
IR:
There is a strong absorption at about 1767 cm-1 which is probably C=O (note that's high for a ketone) and at about 1600 and 1500 cm-1 which is probably C=C and at 1200 cm-1 which is probably C-O. There are no other absorptions of note.... no OH, NH, C≡C or C≡N
13C NMR:
The proton decoupled spectrum shows a total of 8 peaks indicating 8 types of
C. By analysis of the chemical shifts, we have a peak at 170 ppm (C=O in the carboxylic acid derivative range). There are 4 peaks between 120-150 ppm that are 4 sp2 aromatic C, and then 3 peaks between 20-35 ppm that are
most likely from an sp3 hydrocarbon portion.
1H NMR:
The proton spectrum shows a total of 5 peaks indicating 5 types of H.
|
d/ppm
|
multiplicity
|
integration
|
Inference
|
| 7.25 |
d |
2 |
2 x ArH |
| 7.03 |
d |
2 |
2 x ArH |
|
3.05
|
septet
|
1
|
CH coupled to 6H, slightly deshielded |
| 2.30 |
s
|
3
|
CH3, slightly deshielded |
|
1.33
|
d
|
6
|
2x CH3 coupled to 1H
|
(q = quartet, t = triplet, d = doublet, s = singlet)
The most significant structural information
from this are:
- adding integrals shows 14H (or a multiple of)
- since there are 4 ArH, it is a disubstituted benzene, C6H4
- there is a -CH3 group
Summary....
The MS indicated MW = 178
The IR showed the presence of C=O
13C NMR shows 8 C types including C=O and 4 ArC.
H NMR gives disubstituted benzene, C6H4, -CH3, -CH and 2 x -CH3 groups
This information suggests a molecular formula = C11H14O
which matches the MW = 162 (missing 16 = O)
Therefore, molecular formula = C11H14O2
and has an IHD = 5 consistent with the C=O and the benzene system.
Altogether...
|
The fragments we have are:
The IR, 13C and molecular formula imply that the C=O carbonyl is an ester.
The H-NMR 1H septet at 3.05ppm and 6H doublet at 1.33ppm indicate an isopropyl group, -CH(CH3)2
Since the 13C-NMR tells us that there are 4 types of ArC and the H-NMR tells us that there are 2 types of ArH, then the benzene must be para (i.e. 1,4-) substituted (ortho & meta would have 6 types of ArC).
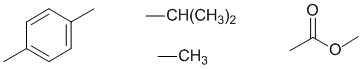
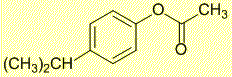
Given that H-NMR shifts of the -CH3 and -CH, the ester must be a phenoxy rather than alkoxy.
The H-NMR chemical shift of the -CH3 of 2.3 ppm and -CH of 3.05 ppm is most consistent with the -CH3 group attached to the C=O and the -CH(CH3)2 on the benzene.
|
|
The final step should always be to check what you
have drawn. The easiest thing to check is usually the coupling patterns you
would expect to see, and the chemical shifts of each unit. You should
be asking yourself : "Does my answer give me what the H-NMR shows ?"
Common errors:
MS incorrectly read the MW for the identified M+ (check the scale). Even, no N.
IR missed the C=O
13C NMR ignored the specifics of the C=O range (i.e. subtype :170 ppm < 190 therefore it's a carboxylic acid or derivative)
H NMR did not get the number of ArH (there are 2+2=4) from the integration, had hydrocarbion pieces that did not fit the integration and coupling patterns
General
- did not check MW with data from MS
- wrong type of C=O (look at 13C NMR C=O range and check IR frequency)
- did not draw a disubstituted benzene
- had the wrong Ar substitution pattern (how many ArH? How many ArH/C types in NMR ?)
- did not check HNMR chemical shifts to correct locate the alkyl groups
- drawing structures that broke the rules of valence!