
Part 5: MECHANISMS
Note that no other reagents are needed in order to complete any of these sequences, you should only be using what is there.
General common errors:
(1) incorrect formal charges, (2) backwards arrows, (3) not showing the arrows for all the bonding changes (i.e. missing steps), (4) misuse of resonance arrows, (5) vague arrows e.g. not clearly starting where the electrons are i.e. on a bond or lone pair, (6) if using "B" to represent a base in the scheme not then defining what B actually is, (7) expecting an alkoxide ion to be present in an acidic environment, (8) not separating the discrete steps.
A1

Common errors : (1) not protonating the carbonyl (2) showing the basic mechanism rather than acidic.
A2
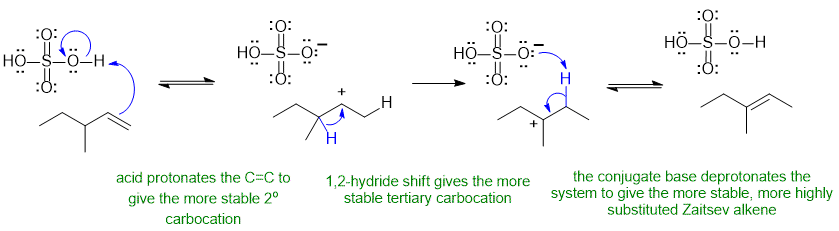
Common errors : not reforming the acid catalyst, missing the carbocation rearrangement
B1
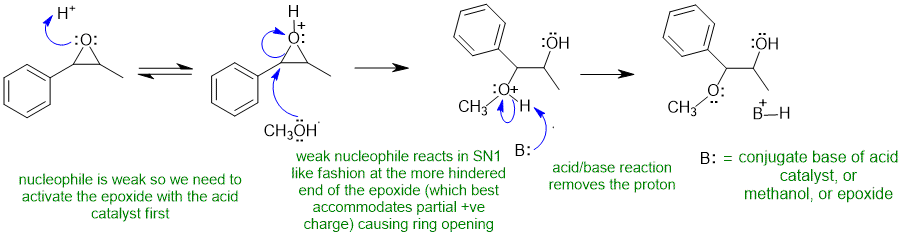
Common errors : (1) not activating the epoxide by protonating prior to attack by the weak nucleophile, (2) incorrect nucleophile (it should be the neutral CH3OH), (3) incorrect regiochemistry (SN1 like at benzylic position due to stabilisation of partial +ve charge).
B2
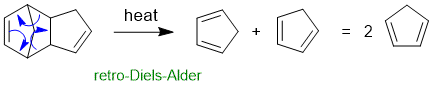
Common errors : (1) not activating the epoxide by protonating prior to attack by the weak nucleophile, (2) incorrect nucleophile (it should be the neutral CH3OH), (3) incorrect regiochemistry (SN1 like at benzylic position due to stabilisation of partial +ve charge).