Answers to the synthesis problems are given below. These answers have been selected as they are short and efficient, but there are probably other reasonable solutions. Note some targets have specific stereochemistry that needs to be considered.
When planning syntheses, one should try to work backwards by functional group analysis. The diagrams show the "retrosynthesis" - the design or plan and then below that the reaction scheme step-by-step (as required in the question). Red arrows try to show carbon-carbon bond forming reactions, blue arrows are functional group manipulations. The blue text rationalise the retrosynthetic analysis and information about important reactions are given in green.
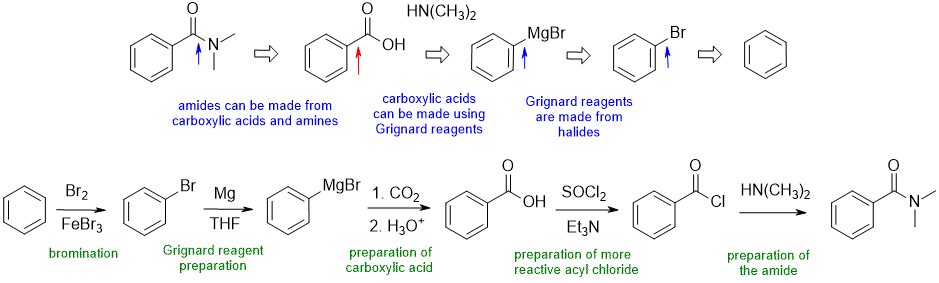
A1

Common errors : (1) attempting to use Friedel-Crafts reactions to make the acid or amide directly
A2
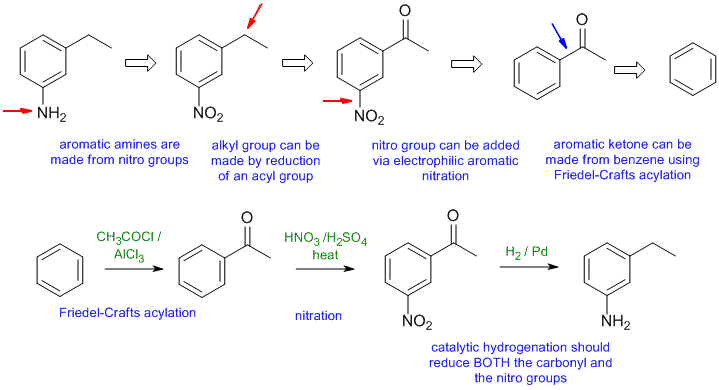
Common errors : (1) order of reactions, (2) attempting to use Friedel-Crafts on nitrobenzene (deactivated), (3) incorrect regiochemistry
B1
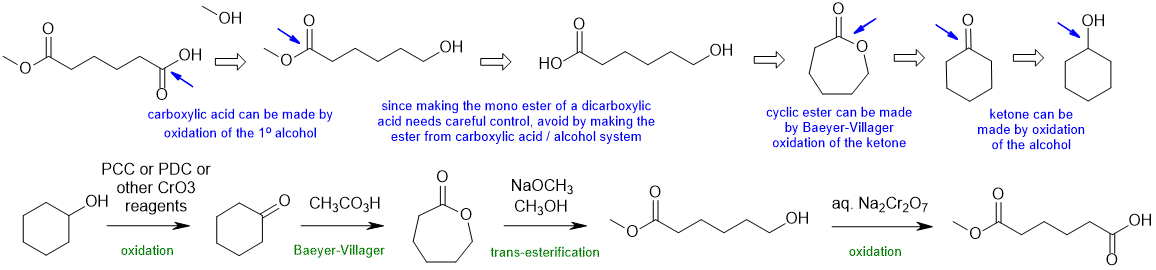
Common errors : (1) attempting to make the mono-ester from a symmetrical dicarboxylic acid, (2) trying to use an acetal to protect an acid (they are typically for aldehydes and ketones)
B2
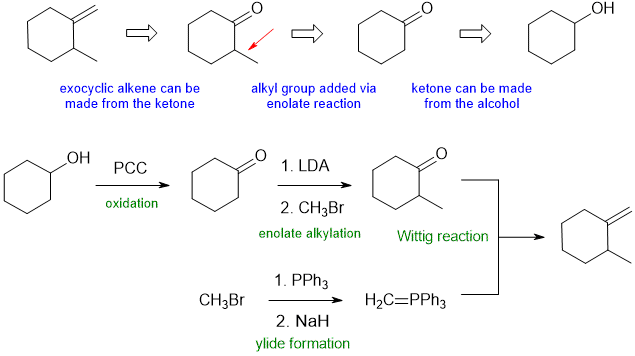
Common errors : (1) missing reagents (e.g. base to form the ylide or enolate), (2) not showing where the ylide (Wittig reagent) came from, (3) ignoring Zaitsev's rule for alcohol elimination
General comments.... Some students started from hydrocarbons with 4 C or less and then made errors in making the materials they could have just used as starting materials.
Common errors included:
(1) incorrect counting of C atoms
(2) trying Friedel-Crafts reactions on deactivated systems
(3) trying to use Friedel-Crafts to directly make acids or amides (it can only be used to give aromatic ketones)
(4) trying to do Grignard reactions in the presence of acidic H atoms (e.g. -OH / -NH or CO2H groups)
(5) trying to make Grignard reagents from R-H rather than R-X (X = Cl, Br or I)
(6) poor management / control / incorrect regio and/or stereochemistry
(7) pKa issues....i.e. using strong nucleophiles (i.e. strong bases) in the presence of acidic groups.