Note that these types of questions are often the most difficult on the examination. This is because they are holistic in that they require a good understanding of the reactions and the concepts and the ability to apply those concepts from the course as a whole. They can't be memorised, they require understanding and ability to apply. Hence they help discriminate those who truly understand from those who have memorised and will soon forget.
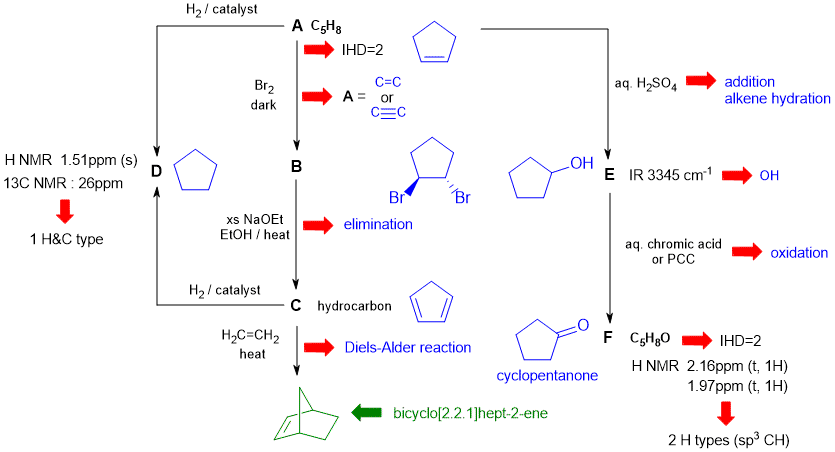
A schematic of the solution is shown below. The information from the question is given in black. Deductions directly from this information are indicated by red arrows. Points that provide potential key information are shown in green which leads to the structures....
In this problem key steps to success are:
1. Recognising the Diels-Alder reaction and hence that C is cyclopentadiene
2. Using the NMR data to deduce D.
3. That the compounds are cyclopentane derivatives
There are, of course, other possible thought pathways.
Common errors:
1. Not recognising that D needed to be cyclic based on the NMR data.
2. Incorrect structure for the bicyclo[2.2.1]hept-2-ene
3. Not knowing the details of some of the reactions involved