- Functional group is an amine, therefore suffix = -amine
- There are two amines, so insert the multiplier di
- The longest continuous chain is C2 therefore root = eth
- Locants for -NH units are 1- and 2-
1,2-ethyldiamine
or
ethyl-1,2-diamine
|

H2NCH2CH2NH2 |
|
- Functional group is an amine, therefore suffix = -amine
- There are two amines, so insert the multiplier di
- The longest continuous chain is C3 therefore root = prop
- Locants for -NH units are 1- and 3-
- There is a C1 substituent = methyl
- The methyl group is located on the amine, so
locant = N
N-methyl-1,3-propyldiamine
or
N-methylpropyl-1,3-diamine
|
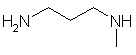
H2NCH2CH2CH2NHCH3 |
|
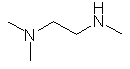
- Functional group is an amine, therefore suffix = -amine
- There are two amines, so insert the multiplier di
- The longest continuous chain is C2 therefore root = eth
- Locants for -NH units are 1- and 2-
- There is are three C1 substituents = trimethyl
- The methyl groups are located different amines,
so locants = N-, N-
and N'-
N,N,N'-trimethyl-1,2-ethyldiamine
or
N,N,N'-trimethylethyl-1,2-diamine
|
(CH3)2NCH2CH2NHCH3 |
|