| Chapter 1: Where are the Electrons ? |
| Chapter 1: Where are the Electrons ? |
To find out whether you know all that you need
to know about this topic, you need to try to answer
these questions before you look at the answers!
You may wish to refer to a periodic table
when answering the following questions.
1. How many electrons are there in each of the following : N-
, O2-, F- and C+
?
2. What is the atomic mass unit for bromine ?
3. How many protons and neutons are there in each of the
following
: 1H, 2H, 12C, 16O, 35Cl
and 37Cl ?
1. Which quantum number defines the
energy level of an orbital
for (a) the hydrogen atom and (b) other atoms ?
2. What are the quantum numbers that describe a 3p orbital ?
3. How many occupied valence orbitals are there in Cl and I ?
4. What are the quantum numbers for an electron in a 3s orbital
?
Use the information given in the box below when answering the next three questions:
| (a) [Ar] | (d) 1s22s22p63s23px23py1 |
| (b) 1s22s22p63s1 | (e) 1s22s22p63s23px13py13pz1 |
| (c) 1s22s22p63p1 |
1. Which of the electron
configurations above best describes the
ground state electron configuration of Cl-?
2. Which of the electron configurations above is possible for
the ground state of P?
3. Which of the electron configurations above describes an
excited
state of Mg+?
4. Is the following electron configuration consistent with Hund's rule of maximum multiplicity?
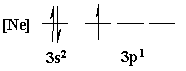

7. What is the orbital energy diagram that best describes the valence electron configuration of O?