|
|
|
|
Friedel-Crafts Acylation of Benzene
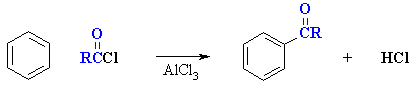
Reaction type: Electrophilic Aromatic Substitution
Summary
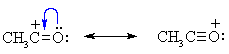
Summary of Limitations of Friedel-Crafts acylations:
|
|
|
|
Step 1:
Step 2:
Step 3:
Step 4: |

|
| © Dr. Ian Hunt, Department of Chemistry |