| Chapter 1: Structure Determines Properties |  |
| Chapter 1: Structure Determines Properties |  |
Answers to the Questions: Electron Configurations
Use the information given in the box below when answering
the next three questions:
| (a) [Ar] | (d) 1s22s22p63s23px23py1 |
| (b) 1s22s22p63s1 | (e) 1s22s22p63s23px13py13pz1 |
| (c) 1s22s22p63p1 |
1. Which of the electron configurations above best
describes the ground state electron configuration of Cl-? (a)
2. Which of the electron configurations above is possible for the ground
state of P? (e)
3. Which of the electron configurations above describes an excited state
of Mg+? (c)
4. Is the following electron configuration consistent with Hund's rule of maximum multiplicity?
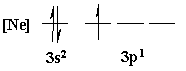
Yes, all electrons unpaired before pairing
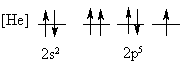
No, two electrons in a p orbital have the same four quantum numbers
7. What is the orbital energy diagram that best describes the valence electron configuration of O ?
Did you draw the core or did you draw the valence electron configuration by mistake ?
Did you draw the valence or did you draw the core electron configuration by mistake ?