Part 6: SYNTHESIS
Answers to the synthesis problems are given below. These answers have been selected as they are short and efficient, but there are probably other reasonable solutions. Note some targets have specific stereochemistry that needs to be considered.
When planning syntheses, one should try to work backwards by functional group analysis. The diagrams show the "retrosynthesis" - the design or plan and then below that the reaction scheme step-by-step (as required in the question). Red arrows try to show carbon-carbon bond forming reactions, blue arrows are functional group manipulations. The blue text rationalise the retrosynthetic analysis and information about important reactions are given in green.
Common errors: incorrect selectivity (regio or stereo) for common reactions (e.g. Markovnikov or anti-Markovnikov OR syn or anti). Miscounting C atoms. Using starting materials that were not hydrocarbons as the question specifically stated. Suggesting reactions of alkanes other than radical halogenation, for example, you can't make alcohols directly from alkanes.
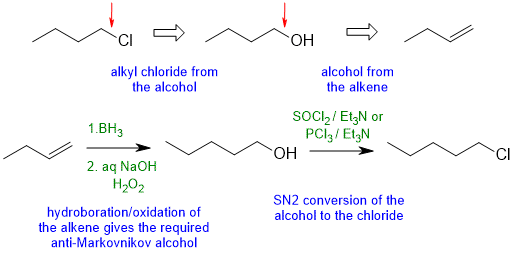
5.1A

Common errors : HCl does not undergo radical addition to give the anti-Markovnikov product. Poor control of the regiochemistry, errors with reagents to convert R-OH to R-Cl via an SN2 reaction.
5.1B
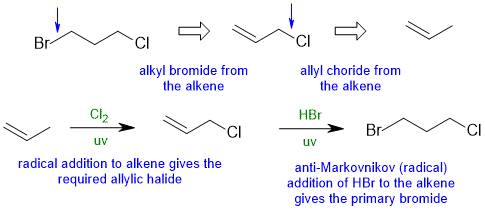
Common errors : Poor contol of the regiochemistry.
5.2A

Common errors : Poor contol of the regiochemistry.
5.2B
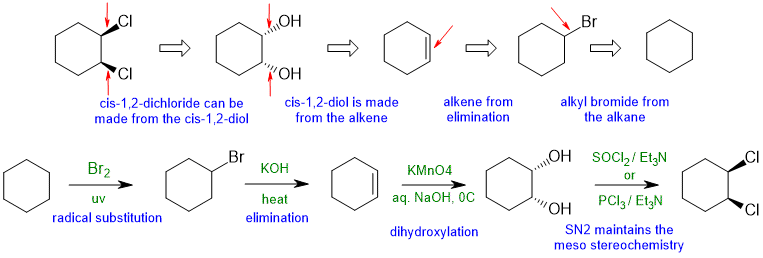
Common errors : Poor contol of the stereochemistry (Cl2 adds to C=C via an anti addition to give a trans-1,2-dichloride).
5.3A
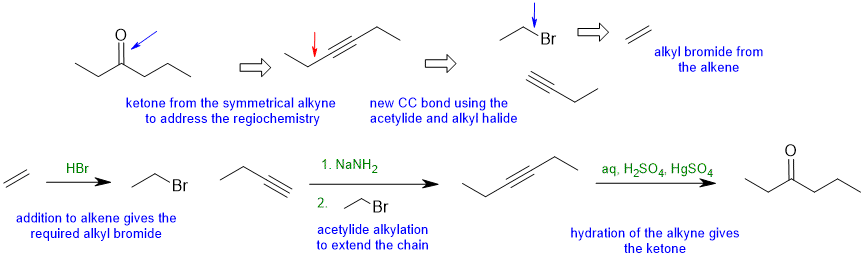
Common errors : Poor contol of the regiochemistry, not showing the synthesise of akyl halides (read the question!)
5.3B

Common errors : Poor contol of the stereochemistry, not showing the synthesise of akyl halides (read the question!) to make the required alkene.