
(7 marks for correct structures, curly arrows, formal charges)
Part 6: MECHANISM
The following diagram shows the solution to the mechanistic question. Note that all the information applies to a single reaction sequence that has been completely described verbally. There is no need for extra reagents or extra steps etc. The curly arrows are drawn specifically to match the text in the question. The biggest problem students have is making sure they understand the language of chemistry. Most students have trouble because they can't draw the structures from the IUPAC names (that means they don't know their nomenclature well enough). Read the words carefully, and then make the curly arrows tell that same story. There is NO need for extra steps. Remember curly arrows go from electron rich to poor and to balance the formal charges at each step - errors on formal charges were common.
a.

(7 marks for correct structures, curly arrows, formal charges)
If you struggled with this part of the question, first draw the compounds whose names were provided, then think about the types of reactions (e.g. acid / base) and try to fill in the structures in the gaps, then finally add the required curly arrows to account for all the bonding changes.
In the image shown above note (a) how the charges balance at each step, and (b) how each of the arrows start and end very close to the structures.
b.
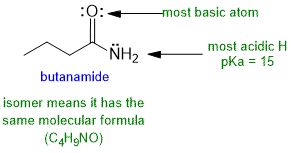
(3 marks : 1 for the structure, 1 for most acidic H, 1 for correct pKa)
c. The most acidic H in the amide are those on the N because (i) N is an electronegative atom and (ii) due to the proximity to the C=O, the conjugate base will be stabilised by resonance (note the similarity of amine and amide pair to alcohol and carboxylic acid).
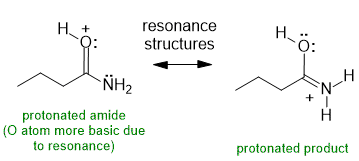
(3 marks :1 for each of the two protonated structures, 1 for the stating the structural relationship)
Common errors: